Pharmacies engaged in non-sterile compounding must undertake a risk assessment to identify the appropriate level (A, B or C) of precautions required to provide adequate protection for personnel and to minimize the risk of contaminating the preparation. This article focuses on the protection of personnel and the use of personal protective equipment (PPE) as described in the NAPRA Model Standards for Pharmacy Compounding of Non-Sterile Preparations and accompanying Guidance Document.
The standards define PPE as “all garb and accessories, such as mask, gloves, gown and safety goggles, that protect the non-sterile preparation and the worker. PPE enables compliance with the expected specifications of a controlled environment and protects the worker from exposure to physical or chemical risks.”
For all levels of compounding, personnel must be trained in the pharmacy’s policies and procedures for conduct, which must include wearing a clean laboratory coat that is reserved for compounding (or a disposable gown) and powder-free gloves. The Master Formulation Record for a particular preparation must indicate the PPE required for compounding and indicate if it is being used as a risk mitigation measure for compounding small, occasional quantities of a potentially hazardous ingredient. This documentation should specify the potential risks to personnel and the extra steps that must be taken to mitigate the risks along with supporting references.
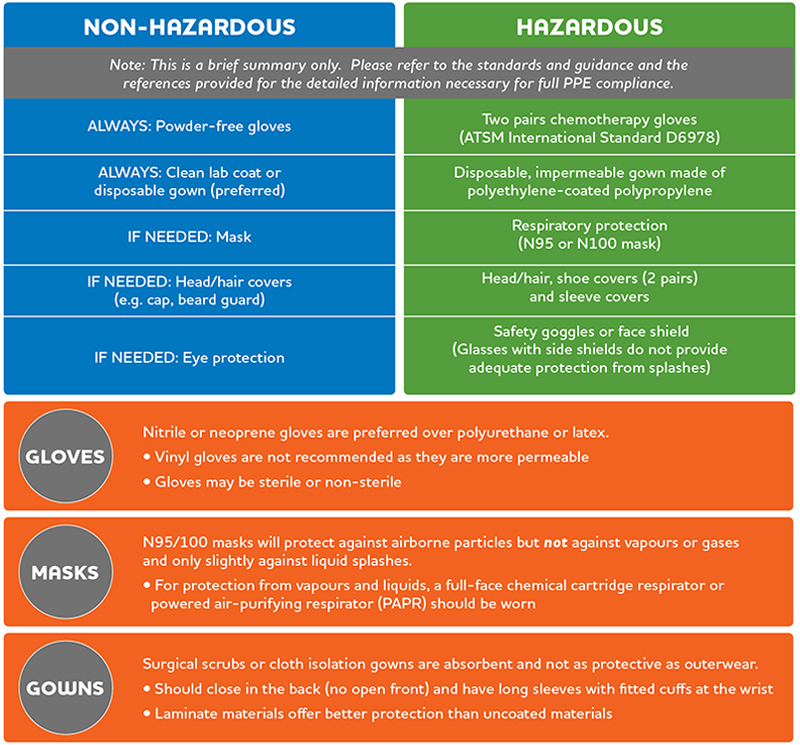
As explained in Section 9 of the NAPRA guidance document, the compounding of products classified as hazardous by NIOSH†, or as irritating to the respiratory tract, skin and mucous membranes by WHMIS‡, requires additional safety measures to protect personnel from chemical exposure.

NIOSH† created a “hierarchy of controls” diagram depicting the various levels of controls that can be implemented to protect personnel by controlling exposure to occupational hazards. It is important to understand that, as a risk mitigation measure, PPE is the least effective compared to other measures, such as engineering controls like ventilated containment devices or closed-off, ventilated rooms. Therefore, personnel must be trained in the proper use of PPE and their work routinely observed and assessed to ensure compliance with PPE procedures. This applies to compounding personnel as well as individuals involved in routine cleaning of the compounding area and/or dealing with spills.
Like any other equipment used in compounding, PPE must be maintained and inspected for integrity before and after each use. Equipment that is damaged or worn must be repaired or replaced to ensure its effectiveness. Section 9.2.3 of the guidance document describes PPE in detail, including gloves, gowns, respiratory protection and more.
Risk mitigation is a key element of implementing the standards and information related to PPE can be found throughout the guidance document. It is important to read and use these documents in their entirety for comprehensiveness and to achieve full implementation.
ADDITIONAL REFERENCES
- Article — Timelines Announced For Non-Sterile Compounding Standards; Pharmacy Connection (Winter 2019) (http://pharmacyconnection.ca/non-sterile-compounding-winter-2019/)
- OSH Answers Fact Sheets; Canadian Centre for Occupational Health and Safety (CCOHS) (https://www.ccohs.ca/oshanswers/about.html)
- Safe Handling of Hazardous Drugs in Healthcare, 2013; Public Services Health & Safety Association (https://www.pshsa.ca/wp-content/uploads/2013/11/PSHSA-Whitepaper-Safe-Handling-of-Hazardous-Drugs-in-Healthcare.pdf)
- Table 5 in List of antineoplastic and other hazardous drugs in healthcare settings, 2016; National Institute for Occupational Safety and Health. (https://www.cdc.gov/niosh/topics/antineoplastic/pdf/hazardous-drugslist_2016-161.pdf)
- Prevention guide—safe handling of hazardous drugs, 2008: Association paritaire pour la santé et la sécurité du travail du secteur affaires sociales (ASSTSAS). (https://asstsas.qc.ca/sites/default/files/publications/documents/Guides_Broch_Depl/GP65A_hazardous_drugs.pdf)
†National Institute for Occupational Safety and Health; Centers for Disease Control and Prevention
‡Workplace Hazardous Materials Information System