When regulatory changes came into effect on January 1, 2023 authorizing pharmacists to prescribe medications for a list of 13 minor ailments, patients immediately took notice. The ability to visit a local pharmacy and be treated for any of the 13 common conditions made it faster and easier to access care—and the data below from the first several months of 2023 show just how many patients took advantage of their pharmacist’s expanded scope.
According to Ministry of Health data, 244,630 minor ailment assessments were performed from January 1 through May 31, 2023, resulting in 205,150 prescriptions. Eighty-five percent of all Ontario community pharmacies participated in prescribing for minor ailments, with at least one pharmacy in every Public Health Unit.
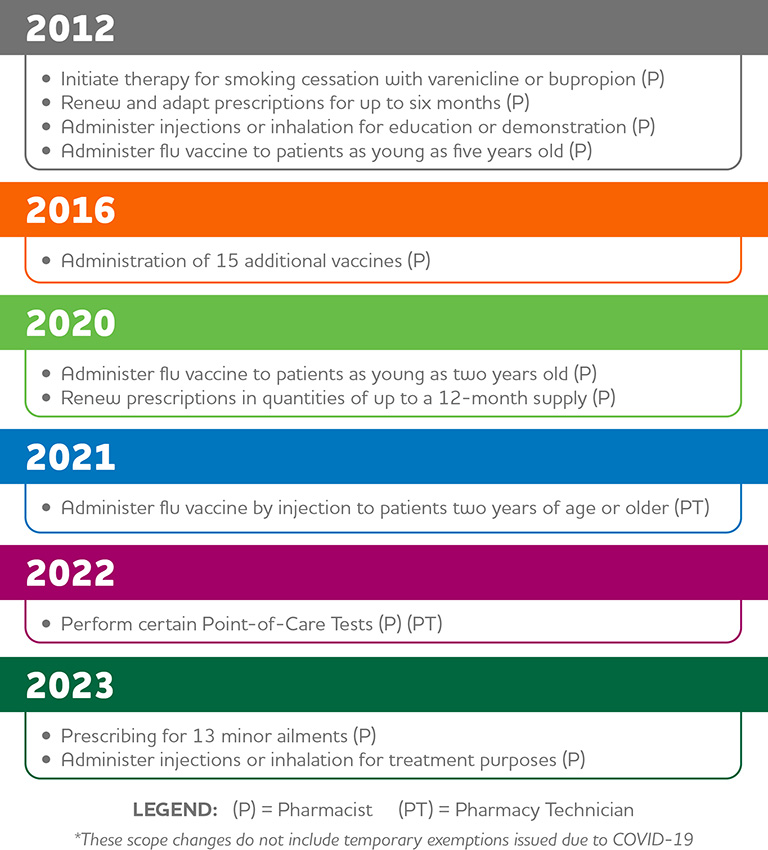
But while minor ailments prescribing was clearly welcomed by patients and most pharmacists, this is just the latest in a series of expanded scope activities dating back more than a decade that continues to evolve in Ontario.
A Timeline of Scope of Practice Changes in Ontario*
Injection and Inhalation updates
As of July 1, 2023, regulatory changes authorize Part A pharmacists, registered pharmacy students and interns to administer substances listed in the regulations by injection and inhalation for purposes other than patient demonstration and education.
This expansion of scope enables pharmacy professionals with injection training to better support their patients with the routine injection or inhalation of medications. This will benefit patients being treated for conditions including osteoporosis, diabetes, or rheumatoid arthritis who may have difficulties with self-injection or who fear needles, or those who may require inhaled medications for conditions such as asthma.
Six new minor ailments proposed
The College submitted proposed regulatory amendments in May 2023 enabling pharmacists to prescribe for an additional six minor ailments that were previously recommended by the Minor Ailments Advisory Group (MAAG) in 2019. Those minor ailments include:
- Acne (mild)
- Aphthous ulcers (canker sores)
- Diaper dermatitis
- Vulvovaginal candidiasis (yeast infection)
- Pinworms and threadworms
- Nausea and vomiting of pregnancy
Government approval is still required for these additional minor ailments to come into effect, with a planned implementation in Fall 2023.
Expanding Scope to Manage Seasonal Respiratory Illnesses
With a potential surge of influenza, Respiratory Syncytial Virus (RSV) and COVID-19 expected later this Fall, coupled with continued pressures on the Ontario healthcare system, the College’s Board of Directors approved a 60-day public consultation on proposed regulatory amendments that would further expand the scope of pharmacy professionals to administer additional vaccines and offer appropriate treatment options for COVID-19 and influenza. The amendments, if approved by government, would:
- give pharmacists and pharmacy technicians authority administer the Respiratory syncytial virus (RSV) vaccine
- give pharmacy technicians authority to administer vaccines from Schedule 3
- give pharmacists authority to prescribe oseltamivir (Tamiflu)
- remove age specific age restrictions for administration of influenza and other Schedule 3 vaccines by pharmacists and pharmacy technicians
- transition authority for pharmacists and pharmacy technicians to administer the COVID-19 vaccine and for pharmacists to prescribe Paxlovid, from the Regulated Health Professions Act (RHPA), Controlled Acts Regulation (107/96) to the Pharmacy Act, General Regulation (202/94).
Following the public consultation, the proposed regulatory amendments will be submitted to the Ministry of Health unless the Board Chair determines that there are substantive changes required that would necessitate the regulation be brought back to the Board for approval.
Further expansion of minor ailments
The College has also re-engaged the MAAG – now renamed the Scope of Practice Advisory Group — to explore the addition of further minor ailments, including those that may require additional scope of practice expansion to support safe and effective prescribing, and prepare recommendations for Board consideration in the fall of 2023, prior to submission to the Ministry of Health. Information on this work will be communicated with registrants and system partners as it becomes available.
Ongoing updates about expanded scope of practice and minor ailments will be shared in e-Connect and on the College’s website.